On January 19, 2024, the U.S. Food and Drug Administration (FDA) approved the 510(k) application of Cosine® Distal Access Catheter produced by AccuMedical (Beijing) Co., Ltd. The approval information has been officially published in the FDA's premarket notification database.
The FDA approval of the Cosine® Distal Access Catheter signifies that its safety and effectiveness have been recognized by U.S. regulatory authorities, offering patients more efficient and safer treatment options.

The Cosine® Distal Access Catheter is designed for use in interventional procedures, facilitating the delivery of interventional devices in peripheral and neurovascular systems. It features a wide range of sizes, excellent torque control, minimal vascular irritation, and superior radiopacity, making it suitable for diverse surgical approaches.
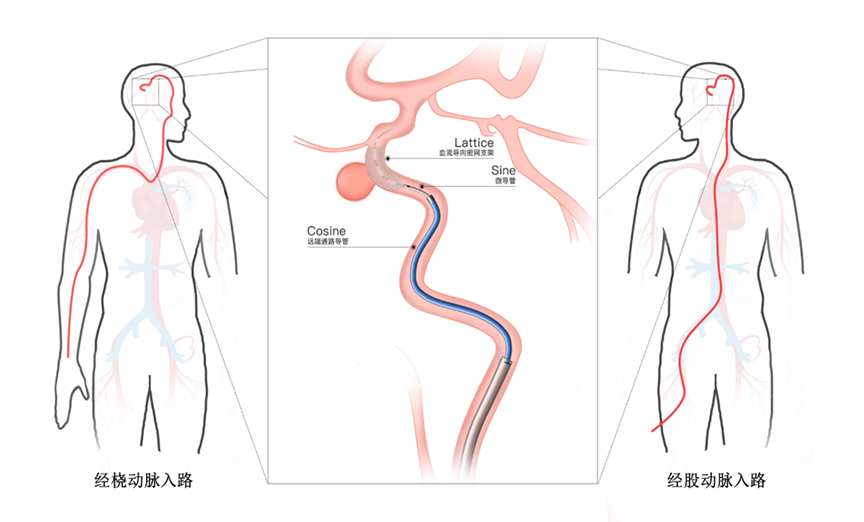
AccuMedical has been dedicated to the neurointervention field for many years. This FDA 510(k) clearance not only opens broader international markets for the company but also provides more comprehensive neurointerventional solutions for global cerebrovascular disease patients and physicians.